|
3/2/2023 0 Comments Sodium atomic radius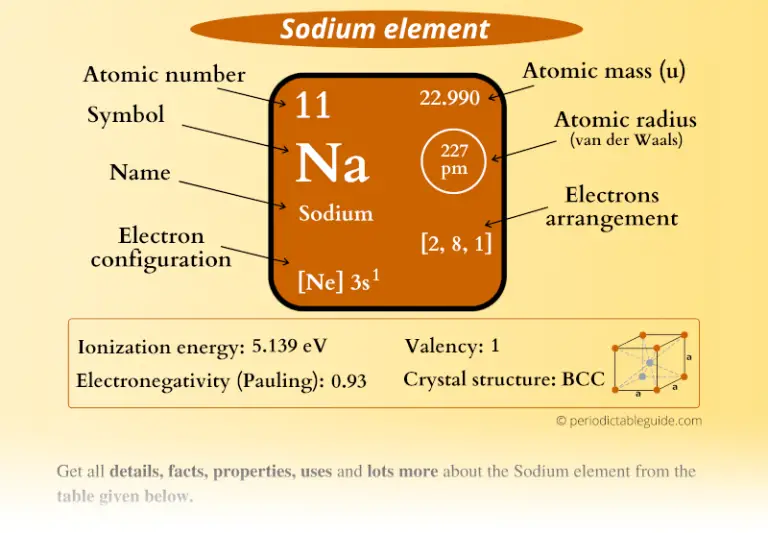
So the one valence electron in sodium feels a certain attraction to the nucleus. If the valence electrons feel a greater attraction they are pulled in closer to the nucleus and the atom gets smaller. Since valence electrons are all in the same level, they feel a greater attraction to the nucleus as we move across the period. Each time we add a proton to a the nucleus the electron (which are negatively charged) feel a greater attraction to the nucleus. Sodium has 11 protons and argon has 18 protons. Remember also that as we move from sodium to argon, the nuclear charge, the number of protons in the nucleus, is also increasing. Remember, and this is important, the valence electrons are in the outer most level of the atom (the furthest from the nucleus). Sodium has one valence electron and as we move across to argon with eight valence electrons. Here is the portion of lecture which develops the table below. To get started we must consider the electron configuration for the eight elements in the third period. To develop an explanation of the trends in atomic radii we need to discuss several new ideas valence electrons, inner core electrons, effective nuclear charge and shielding. 
Here is our lecture discussion on this issue. Cesium has a large number of protons, yet it is one of the largest atoms, not the smallest. That might explain the trend in atomic radii going across a period but it does not explain the trend going down a group. Since electrons are attracted to protons we might expect that the more protons the smaller the radius. 
So we could argue that going from hydrogen through the elements the number of protons increases. We might be able to use that argument to explain the trend within a group, but when we look at the atomic radius trend going across a period the radius decreases. Since electrons repel each other due to the like charges, and the size of the atom will increase. How do we understand these two trends? Explanations of trends in atomic radii that do not work perfectly: We might argue that going from hydrogen through the elements that the number of electrons increase. Also notice that going across a period the atomic radius decreases. Looking at the table above notice from lithium to cesium, going down the group, the atomic radius increases. After collecting large amounts of data generally acceptedatomic radii are known for most elements in the periodic table.Atomic radii determine this way are also called covalent radii.(we'll discuss the term covalent in the next chapter) Atomicradii for metals can be estimated from the distance between metalatoms in the pure solids. Care must betaken in these types of determinations however. Additional distances canbe obtained from other distances between atoms. Therefore theatomic radius of chlorine is 0.994 Å. For example, the distance betweenthe two chlorine atoms in Cl 2 is known to be 1.988 Å.To get the atomic radius we assume the distance between the twonuclei is the sum of two chlorine atomic radii. So some approximations are madein determining this parameter. It is difficult todefine a sharp boundary for distance between the electrons in anyparticular atom and the nucleus. Our quantum mechanical description of an atom suggests avery broad region for finding the electron. The immediate question is what is an atomicradius. The first property to explore isatomic radius. The Quantum Mechanical model of theatom can 'tested' by looking at the experimental data of atomicradius and ionization energy. The strength of any model is in it ability to explainexperimental observations. The first property to explore is atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
